In 2025, a new proficiency test will be conducted to evaluate the yeasticidal activity of disinfectant products. This test, based on the EN 13624:2021 standard, is aimed at laboratories performing disinfectant and antiseptic tests in the medical field. The standard outlines methods and requirements for evaluating products designed for disinfecting hands, surfaces, and instruments.
EN 13624:2021 Standard Overview
EN 13624:2021 is a European standard that defines a quantitative suspension test to assess the fungicidal and yeasticidal activity of chemical disinfectants and antiseptics in the medical sector. This standard focuses on products used for disinfecting hands, surfaces, instruments, and textiles in clinical settings such as hospitals, medical centers, and laboratories. It ensures that disinfectants meet the necessary efficacy standards in critical situations, where disinfection plays a key role in infection prevention.
The scope of the standard covers various medical applications, including hygienic and surgical handwashing, surface disinfection via methods like rubbing, spraying, or flooding, immersion of instruments in disinfectant solutions, and textile disinfection, especially in hospital laundry services. The EN 13624:2021 standard provides guidelines for evaluating two types of antimicrobial activity: fungicidal and yeasticidal.
Fungicidal activity is assessed using conidiospores of Aspergillus brasiliensis, while yeasticidal activity is evaluated with vegetative cells of Candida albicans. These microorganisms are selected due to their medical relevance, particularly their ability to cause severe infections, especially in immunocompromised patients. The upcoming 2025 proficiency test will focus specifically on Candida albicans.
Candida albicans is a clinically significant pathogenic yeast, known for causing nosocomial infections. It is the primary agent of candidiasis, which can range from superficial mucocutaneous infections to invasive forms affecting immunocompromised patients. C. albicans is notable for its adaptability, biofilm formation, and resistance mechanisms, making it a model organism for yeasticidal activity tests, such as those conducted according to EN 13624.
Test Conditions
To ensure accuracy and reproducibility, EN 13624:2021 provides detailed test conditions for evaluating disinfectants. These conditions are essential to reflect real clinical scenarios where disinfectants are used.
Two types of conditions are defined based on the presence or absence of organic matter:
- Clean conditions: Simulate an environment relatively free of organic contaminants. In this case, 0.3 g/L bovine albumin is used as an interfering substance, representing situations such as disinfecting hands or surfaces in pre-cleaned clinical areas.
- Dirty conditions: Simulate a more contaminated environment, where the disinfectant must act in the presence of organic matter. For this, a mixture of 3.0 g/L bovine albumin and 3.0 mL/L erythrocytes is used, reflecting situations where blood or biological residues may be present on surfaces or instruments.
Additionally, the standard specifies parameters such as temperature and contact time. For Candida albicans, test temperatures range between 20°C and 50°C, depending on the product and its application. Contact times vary from 1 to 60 minutes, with shorter times for fast-acting disinfectants, such as those used for hand disinfection, and longer times for surface and instrument disinfection.
Test Methods
The primary method for evaluation in EN 13624:2021 is the dilution-neutralization method, which ensures that the disinfectant stops acting at the precise moment, preventing further antimicrobial activity beyond the specified contact time. The method involves diluting the disinfectant in an appropriate neutralizer to halt its action, which is crucial for obtaining accurate results on the surviving microorganisms.
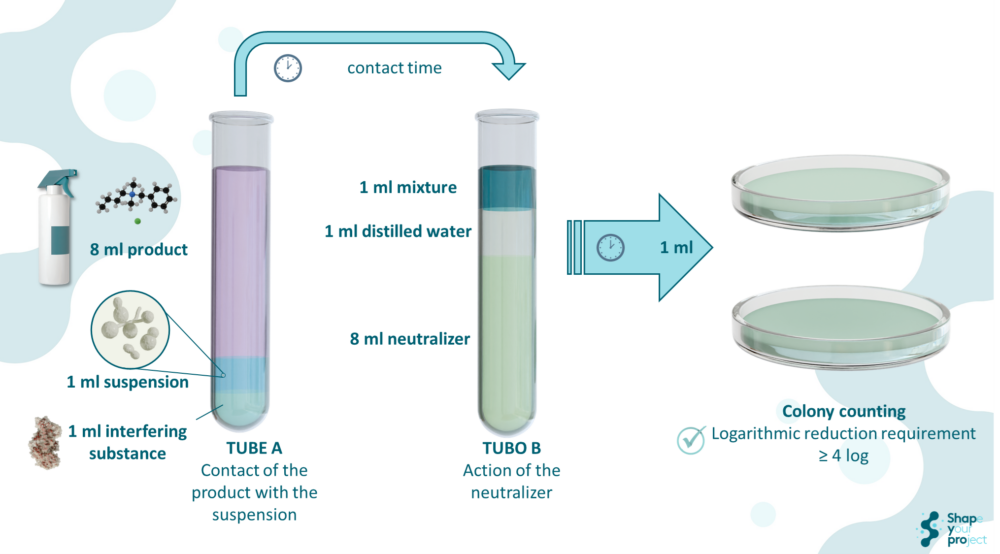
When the dilution-neutralization method is not feasible, the membrane filtration method is used. This method filters the test solution through a membrane that retains microorganisms, allowing for their subsequent counting to assess the reduction achieved by the disinfectant.
A critical aspect of the standard is the validation of the neutralizer. The neutralizer must be pre-tested to ensure it does not affect the viability of microorganisms and can fully halt the disinfectant’s activity without interference. Validation is conducted using specific controls, ensuring the test’s reliability and that results are not altered by other factors.
Efficacy Requirements
To comply with the EN 13624:2021 requirements, disinfectants must demonstrate a minimum 4-logarithmic reduction in viable cells of Candida albicans, equating to a 99.99% elimination of microorganisms. This level of efficacy is crucial in clinical settings, where patient safety depends on the disinfectant’s ability to effectively eliminate pathogens.
Meeting these requirements ensures that disinfectant products are suitable for use in critical medical areas where disinfection serves as a key barrier against infection transmission.
Participate in Our Proficiency Test
The proficiency test will follow the principles established in EN 13624:2021 to evaluate the efficacy of disinfectants against Candida albicans. The test conditions will include the preparation of a Candida albicans suspension, which will be exposed to the disinfectant in a controlled environment, adhering to parameters such as contact time, temperature, and cleanliness conditions according to the standard.
The proficiency test aims to achieve several key objectives to ensure the quality and technical competence of participating laboratories:
- Compliance with accreditation requirements: The test will allow laboratories to meet the requirements of EN ISO/IEC 17025, which mandates regular proficiency testing to ensure technical competence within the accreditation scope.
- Identification of technical issues: Laboratories will have the opportunity to identify any problems in their test methods and take corrective actions to improve accuracy and reliability.
- Establishing method efficacy: The test will provide a detailed assessment of the efficacy of disinfectants against Candida albicans, ensuring they meet the microbial reduction standards established in EN 13624:2021.
- Enhancing customer confidence: Laboratories that participate will receive a certificate validating their competence, providing increased confidence to their clients in the services offered.
- Validation of declared uncertainty estimates: The test will also allow laboratories to validate their uncertainty estimates, a key factor in maintaining the quality of results.
- Ongoing performance monitoring: Proficiency testing is essential for monitoring the continuous technical performance of laboratories, ensuring consistency and quality in results over time.
The disinfectant product to be tested will be supplied by Shapypro, and a detailed protocol will be provided, including specific test conditions such as contact time, temperature, and whether to apply clean or dirty conditions. This protocol will allow laboratories to perform the tests under standardized conditions that reflect practical disinfection scenarios.
The proficiency test will consist of two stages:
- Determination of effective concentration: Participants will calculate the minimum disinfectant concentration required to achieve the necessary reduction of Candida albicans.
- Determination of reduction rate: After determining the effective concentration, participants will evaluate the disinfectant’s reduction rate under the prescribed conditions (based on contact time and temperature).
This proficiency test is designed for laboratories that meet the following profiles:
- Laboratories accredited under EN ISO/IEC 17025.
- Laboratories seeking to improve their technical competence in disinfectant evaluation.
- Laboratories aiming to conduct ongoing performance monitoring to ensure result consistency and quality.
Additionally, Shapypro guarantees that the organization and processing of results will be carried out in accordance with applicable international standards, such as ISO/IEC 17043, ISO 5725, and ISO/IEC Guide 35, ensuring comparability and validity of results across participating laboratories.
For more information on this proficiency test or participation requirements, feel free to contact us at Shapypro.
Why Choose SHAPYPRO’s Proficiency Tests?
With over 10 years of experience in evaluating the efficacy of disinfectant products, we offer proficiency testing programs that comply with the requirements of ISO/IEC 17043 and conform to EN 14885:2022/AC:2023.
We are the only accredited provider worldwide in this field of testing under ISO/IEC 17043. [Click here to download the accreditation]
Our extensive knowledge allows us to assist clients in developing new methods and R&D projects. We have previously developed proficiency testing programs under EN 13624.
Additionally, we are manufacturers of soft diluted soap necessary for both developments.
We provide customized proficiency tests. All tests are conducted in accordance with internationally validated standards and guidelines.
Interested in our 5-year schedule? Contact us!


