The effectiveness of disinfectants is a decisive factor in sectors where hygiene, safety, and regulatory compliance are non-negotiable. In Europe, the EN 1656 standard has become a benchmark for demonstrating the bactericidal activity of chemical disinfectants and antiseptics through a standardized quantitative suspension test.
This article highlights the relevance of EN 1656 in the veterinary area, explains how suspension tests work, the importance of Proficiency Testing (PT), and how results reinforce laboratory reliability within the chemical sector, under the framework of Regulation (EU) No 528/2012 on biocidal products.
EN 1656: What It Is And How It Applies
The EN 1656:2009 standard defines a quantitative suspension test to measure the bactericidal effectiveness of chemical disinfectants in the veterinary area. Its application ensures that a disinfectant can significantly reduce bacterial load under controlled conditions.
The standard establishes:
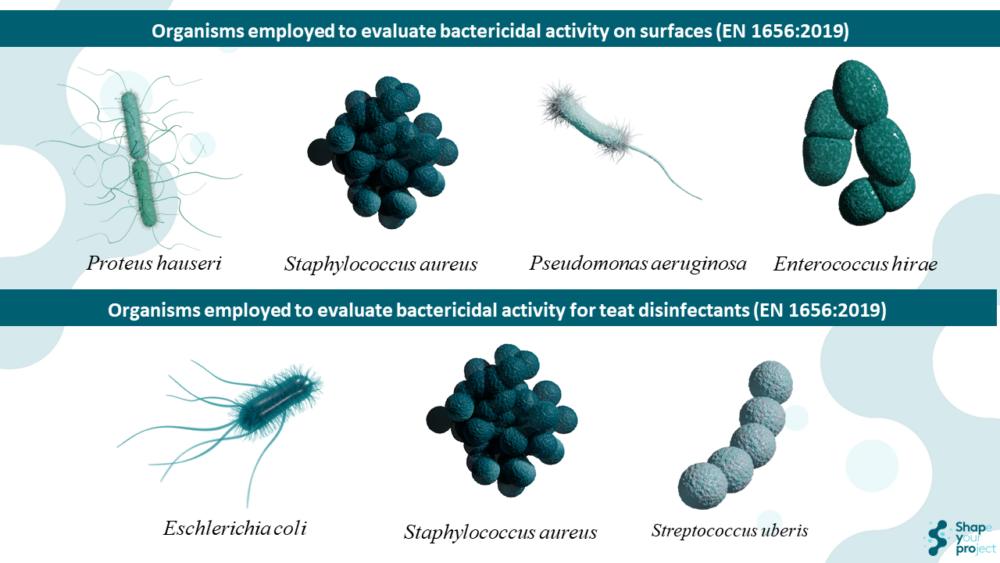
- Reference bacterial strains (e.g., Staphylococcus aureus, Enterococcus hirae).
- Specific experimental conditions (concentration, contact time, temperature).
- Acceptance criteria expressed as logarithmic reduction.
By establishing these parameters, EN 1656 provides a common language among manufacturers, laboratories, and regulatory authorities, allowing for comparable and consistent results across Europe.
Quantitative Suspension: The Core Methodology Of EN 1656
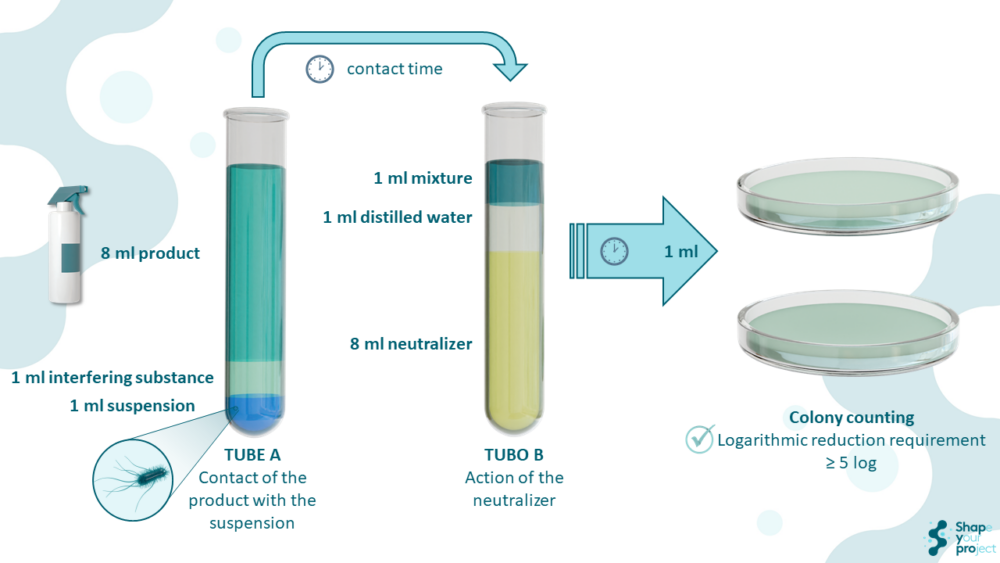
The suspension test includes five key steps:
- Preparation of microbial suspension: Standardized bacterial strains are selected to serve as the microbiological challenge.
- Exposure to the disinfectant: The suspension is mixed with the product at a specific concentration for a defined contact time.
- Neutralization: A neutralizer is applied to stop the disinfectant’s action.
- Bacterial count: Samples are plated, and the number of surviving bacteria is quantified.
- Efficacy calculation: Activity is expressed as a logarithmic reduction. A reduction of ≥ 5 log (99.999% elimination) is generally considered satisfactory.
This provides an objective measurement of disinfectant performance under standardized conditions.
Application Of EN 1656 In The Veterinary Area
The method sets the minimum requirements for bactericidal activity of chemical disinfectants and antiseptics that remain homogeneous and stable when diluted with hard water, or with water in the case of ready-to-use products.
- Products are tested at a concentration of 80% or less, as dilution always occurs when adding microorganisms and interfering substances.
- It applies to equipment disinfection by immersion, surface disinfection by wiping, spraying, flooding, or other means, and teat disinfection in veterinary contexts — including breeding, husbandry, production, veterinary care facilities, animal transport, and disposal (except when animals enter the food chain after death).
- It is not applicable to hand hygiene products, which are covered by EN 14885.
This corresponds to a Phase 2, Step 1 test, designed to reflect activity under practical conditions.
Proficiency Testing (PT) Under EN 1656
Although the standard provides a clear protocol, laboratories must validate the reliability of their results. This is where Proficiency Testing (PT) plays a key role.
Participation in PT programs offers:
- Interlaboratory comparison, ensuring consistency.
- Error detection, whether methodological or technical.
- Competence recognition, demonstrating compliance with ISO/IEC 17043.
- Continuous improvement, based on objective outcomes.
At SHAPYPRO, we design international PT programs focused on EN 1656, helping laboratories in the chemical sector demonstrate their technical competence at a global level.
Evolution Of EN 1656 In Europe
The EN 1656 standard, developed by CEN-CENELEC, is periodically reviewed to remain aligned with new scientific challenges such as resistant bacteria and sustainable product development. Its application is fully integrated into the legal framework of Regulation (EU) No 528/2012 on biocidal products.
The Role Of SHAPYPRO In Applying EN 1656
At SHAPYPRO, we support laboratories and manufacturers in implementing tests under EN 1656. Our Proficiency Testing programs accredited to ISO/IEC 17043 strengthen result reliability and reinforce trust among clients and authorities.
For further details, you can read our article on the comparison of suspension tests for disinfectants or visit our contact page.
Conclusions
The EN 1656 standard is essential for validating the bactericidal efficacy of chemical disinfectants and antiseptics in the veterinary field.
Its application ensures reliable and comparable results, while Proficiency Testing enhances quality, continuous improvement, and international recognition.


