Non-conformity is not an anomaly to be hidden or avoided in quality management systems. On the contrary, it is a key indicator of how well an organisation controls its processes, complies with requirements, and responds to risk. Correctly identifying, analysing, and addressing non-conformities is a cornerstone of continuous improvement and regulatory compliance.
In regulated sectors such as laboratories, cosmetics, biocides, and industrial testing, the improper management of non-conformities can lead to audit findings, loss of accreditation, regulatory actions, or compromised data integrity. This article explains what a non-conformity is, how it should be managed, and why root cause analysis is critical to ensuring effective and sustainable corrective actions.
What Is A Non-Conformity
A non-conformity (NC) is any deviation from a specified requirement. These requirements may originate from:
- International standards (e.g. ISO/IEC 17025, ISO 9001, ISO/IEC 17043)
- Regulatory frameworks
- Internal procedures and SOPs
- Contractual or customer requirements
A non-conformity indicates that a process, result, or activity does not comply with what was planned or required.
It is important to distinguish non-conformities from isolated errors. While an error may be accidental and immediately corrected, a non-conformity reflects a systemic weakness that requires structured investigation and corrective action.
Types Of Non-Conformities
Non-conformities can be classified according to their impact and scope.
Major Non-Conformities
Major non-conformities indicate a significant failure of the management system or a direct risk to the validity of results. Examples include:
- Absence of required procedures
- Invalid or unreliable test results
- Failure to implement corrective actions
- Lack of competence or authorisation
These findings often require immediate action and may affect accreditation or regulatory approval.
Minor Non-Conformities
Minor non-conformities are deviations that do not immediately compromise the system but still require correction. Examples include:
- Incomplete records
- Isolated procedural deviations
- Documentation inconsistencies
Although less severe, repeated minor non-conformities may indicate deeper systemic issues.
Why Non-Conformities Should Not Be Treated As Failures
A mature quality system does not aim to eliminate non-conformities entirely, but to manage them effectively. Non-conformities provide:
- Objective evidence of system performance
- Insight into process weaknesses
- Opportunities for improvement
- Data for risk-based decision-making
Organisations that address non-conformities transparently and systematically tend to improve their robustness and resilience over time.
Root Cause Analysis In Non-Conformity Management
Corrective actions are only effective if they address the root cause of the non-conformity, not just its symptoms.
A root cause is the underlying reason that allowed the non-conformity to occur. If it is not identified and mitigated, the same issue is likely to recur.
Common Root Cause Analysis Methods
Several structured methodologies are commonly used to identify root causes:
The 5 Whys
This technique involves repeatedly asking “why” until the fundamental cause is identified. It is simple and effective for straightforward issues.
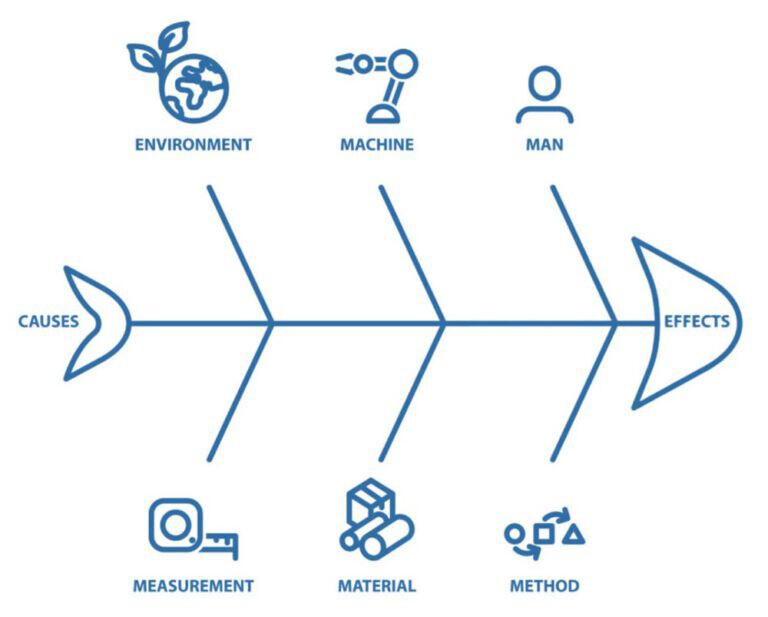
Ishikawa (Fishbone) Diagram
This method categorises potential causes into areas such as:
- People
- Methods
- Equipment
- Materials
- Environment
- Management
It is particularly useful for complex or multifactorial non-conformities.
Failure Mode And Effects Analysis (FMEA)
FMEA is a proactive method that evaluates potential failure points, their causes, and their impact, allowing organisations to prioritise corrective actions based on risk.
Corrective Actions In Response To Non-Conformities
Once the root cause has been identified, corrective actions must be:
- Specific: directly addressing the identified cause
- Proportionate: appropriate to the risk and impact
- Documented: clearly recorded and traceable
- Implemented: effectively applied within defined timelines
Corrective actions may include procedural updates, staff training, equipment maintenance, method validation, or changes to resource allocation.
Verification Of Effectiveness
Corrective action does not end with implementation. It must be verified to ensure that:
- The non-conformity has been eliminated.
- The root cause has been effectively controlled.
- No new risks have been introduced.
Verification may involve audits, trend analysis, repeat testing, or performance monitoring.
Preventive Thinking And Continuous Improvement
Although many standards now emphasise risk-based thinking rather than preventive actions as a separate concept, effective non-conformity management inherently supports prevention.
By analysing trends and recurring issues, organisations can anticipate potential failures and strengthen their systems before non-conformities occur.
The Role Of SHAPYPRO In Non-Conformity Management
In highly regulated environments, managing non-conformities requires technical expertise, regulatory understanding, and structured methodologies. SHAPYPRO supports laboratories and organisations by:
- Assisting in non-conformity analysis
- Supporting root cause investigations
- Reviewing corrective action plans
- Aligning actions with applicable standards and regulations
- Strengthening quality systems through continuous improvement
This approach ensures that non-conformities are not only corrected, but used as drivers for long-term system improvement.
Conclusion: Turning Non-Conformities Into Improvement
Non-conformities are not signs of failure, but indicators of how effectively a quality system functions. When managed correctly through structured root cause analysis and meaningful corrective actions, they become powerful tools for improvement.
Organisations that treat non-conformities as opportunities rather than obstacles are better positioned to maintain compliance, ensure data integrity, and continuously enhance their technical and regulatory performance.


