Nonconformities management is essential to technical excellence. Technical excellence is not only measured by the precision of results but also by the ability to detect, analyze, and correct deviations that may arise during operations.
Within its Quality Management System, accredited under ISO/IEC 17043:2023, SHAPYPRO has developed and updated its procedure for the management of nonconformities, deviations, and incidents, a key document that ensures traceability, transparency, and continuous improvement across all its processes.
The most recent version of the procedure (V03, September 2025) reinforces digitalization, collaborative management, and systematic control, consolidating a quality model based on evidence, prevention, and accountability.
Nonconformities Management in the ISO/IEC 17043 Framework
This procedure is a core element of SHAPYPRO’s quality system.
Its purpose is to ensure that every nonconformity (NC), deviation (DV), or incident (IN) is managed in a structured, documented, and effective manner—from detection to the closure of corrective and preventive actions.
The ISO/IEC 17043:2023 standard, which governs the competence of Proficiency Testing (PT) providers, requires accredited bodies to maintain documented procedures for handling nonconforming events.
In this context, this procedure guarantees that each occurrence is evaluated through objective, traceable, and measurable criteria, enhancing confidence in SHAPYPRO’s impartiality and the validity of its results.
Furthermore, this procedure is fully integrated with other essential documents within SHAPYPRO’s Quality Management System, including document and record control, internal audits and corrective actions, and risk and opportunity management.
From Detection to Action in Nonconformities Management
The main objective is to establish a standardized methodology for identifying, investigating, and resolving any event that may affect the quality or technical integrity of SHAPYPRO’s services.
Key functions include:
- Detection and registration of nonconformities, deviations, and incidents.
- Root cause analysis using structured problem-solving tools.
- Implementation of corrective and preventive actions to eliminate the identified causes.
- Verification of the effectiveness of actions before closure.
- Documentation and traceability of all activities to promote continual improvement.
This procedure applies to both technical activities (Proficiency Testing schemes, sample preparation, data analysis) and administrative processes (communication, document control, and customer feedback).
Classification Of Nonconformities: Critical, Major, And Minor
A three-tier classification system for nonconformities, allowing the organization to prioritize resolution according to the severity and potential impact on quality.
Types of nonconformities:
- Critical: Events that compromise result validity or ISO/IEC 17043 compliance.
Example: errors in PT sample preparation or result traceability. - Major: Significant deviations requiring immediate action but not affecting result integrity.
Example: partial noncompliance with a procedure or missing data in documentation. - Minor: Isolated or administrative issues that do not impact technical quality but must be corrected.
Example: delayed approval or misfiled documentation.
This structured classification ensures that each case is addressed with the appropriate level of depth and urgency.
Digital Tools in Nonconformities Management
All events are recorded in standardized digital forms — Annex I (NC/DV/IN Register) and Annex II (Corrective Action Plan) — within SHAPYPRO’s internal quality management platform.
This system allows for:
- Immediate assignment of responsibilities for investigation and follow-up.
- Chronological tracking of actions, comments, and decisions.
- Automated alerts for pending reviews and closure deadlines.
- Statistical trend analysis to identify recurring patterns and improvement areas.
Digital record management not only enhances traceability but also supports SHAPYPRO’s commitment to sustainability by minimizing paper use and streamlining document review and approval workflows.
Root Cause Analysis And Corrective Actions
The methodological foundation relies on root cause analysis, using approaches such as the “5 Whys” or the Ishikawa (fishbone) diagram, depending on the complexity of each event.
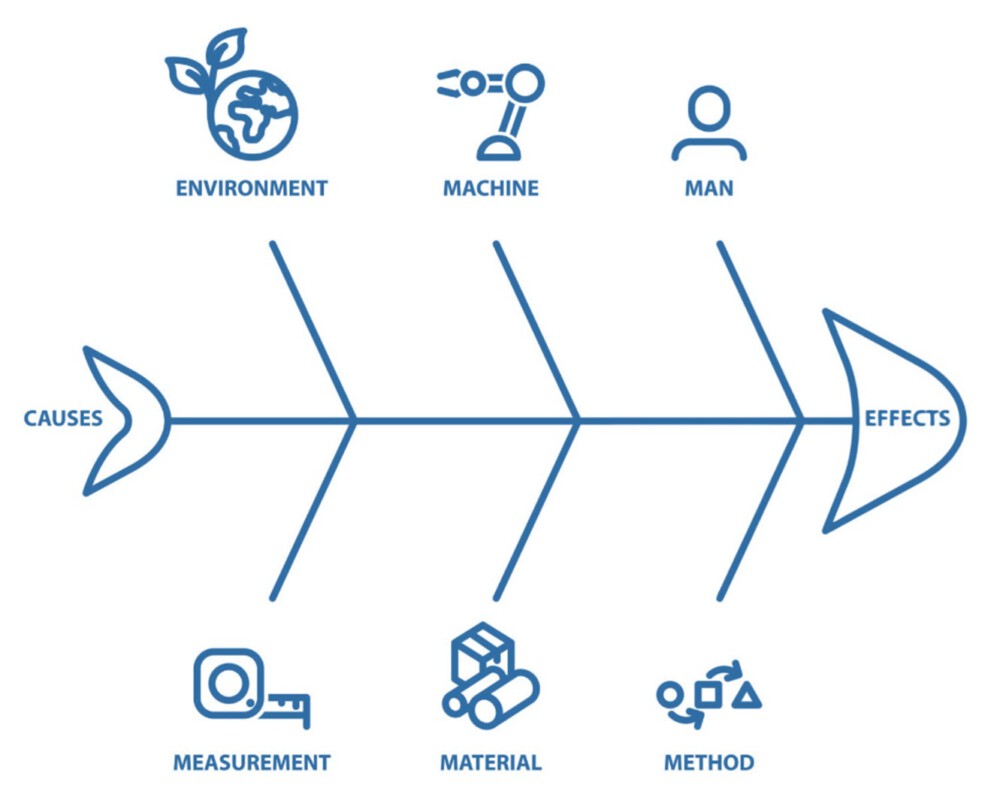
The goal is not merely to correct a deviation but to identify its underlying cause to prevent recurrence.
Each investigation includes:
- Detailed description of the event.
- Identification of immediate and root causes.
- Evaluation of the real and potential impact.
- Corrective action plan with responsibilities and deadlines.
Types of actions:
- Immediate or containment actions: quick measures to prevent escalation.
- Remedial actions: restore compliance or data integrity.
- Preventive actions: eliminate recurrence through procedural improvement or staff training.
Nonconformities can only be closed once the effectiveness of the corrective action is verified and approved by the Quality Assurance department.
Prevention, Containment, And Continuous Improvement
The management of nonconformities at SHAPYPRO is structured around the PDCA cycle (Plan–Do–Check–Act), ensuring that every deviation drives organizational learning and continuous improvement.
Each NC, DV, or IN is viewed not as a failure but as an opportunity for optimization.
From these analyses, performance indicators are derived and used to refine training programs, risk management, and process controls.
The Technical Direction and Quality Assurance teams hold regular review meetings to:
- Evaluate the status of open nonconformities.
- Identify recurrence or trends.
- Recommend preventive actions and process adjustments.
- Share findings across relevant departments.
This proactive model turns incident management into a catalyst for improvement, rather than a reactive task.
A Living And Continuously Updated System
The internal procedure introduces significant updates, including:
- Consolidation of annexes and digital templates.
- Integration of electronic signature and monitoring tools.
- Clearer definitions of responsibilities and escalation levels.
- Stronger connection with (Risk Management).
These improvements enhance the coherence of SHAPYPRO’s quality system and align it more closely with ISO/IEC 17043:2023 requirements, ensuring agile, traceable, and verifiable deviation management.
Conclusion: Transparent Nonconformities Management
With the objectives to reflect SHAPYPRO’s unwavering commitment to technical excellence, transparency, and accountability.
By implementing this procedure, SHAPYPRO ensures that every nonconformity is effectively managed, minimizing its impact and reinforcing confidence among clients, accreditation bodies, and partner laboratories.
More than a compliance requirement, represents a dynamic learning tool that fosters continuous improvement and strengthens the reliability of internal processes.
Through this approach, SHAPYPRO demonstrates that quality is not merely about meeting standards — it is built daily through traceability, awareness, and data-driven improvement.
📖 Learn more about our accredited quality management approach:
👉 SHAPYPRO: The Only Provider Accredited For Biocides Proficiency Testing Under ISO/IEC 17043:2023


