Chemical disinfectants and antiseptics should be subjected to efficacy test from their intended area of application.
In our 2024 agenda we have published a summary table from EN 14885
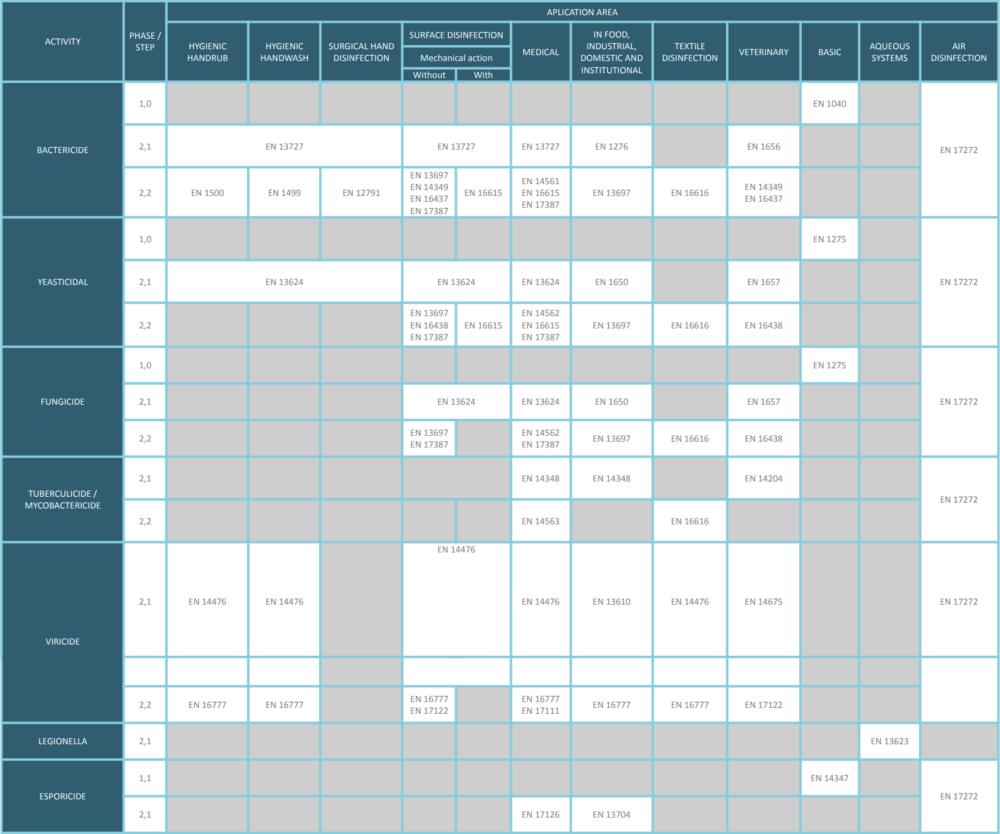
Category of test (procedures for claiming activity)
Today we are focussing on Phase 2, step 2 tests, which are quantitative laboratory tests to establish that a product has bactericidal, fungicidal, yeasticidal, mycobactericidal, tuberculocidal, sporicidal, virucidal or phagocidal, algaecidal or oocysticidal activity when applied to surface or skin under simulated practical conditions.
This type of test provides information about the activity against desiccated microorganisms on inanimate surfaces or on living tissues or against non-desiccated microorganisms on living tissues.
Test shall be carried out under the minimum requirements/obligatory conditions as specified in the standards. According to the claimed use of the product, test under additional conditions (test organisms, temperature, contact time and interfering substances)
Generally, these tests are needed in combination to support efficacy claims for chemical disinfectant or antiseptic. Only in exceptional circumstances deviation from this principle is allowed, both results shall be considered in determination of the label claim.
PROFICIENCY TESTING PERFORMANCE PHASE 2, STEP 2 CONDITIONS
We have developed the first ring trials according to EN 1500 and EN 1499.
| Program of proficiency tests performed | |
| Method | ID |
| EN 1500:2013 | PT-1500-2-0921 |
| EN 1499:2013 | PT-1499-2,2-0223 |
This year 2024 we have planned to develop another phase 2, step 2 conditions efficacy test. In this case, we will coordinate virucidal efficacy testing according to EN 16777 Chemical disinfectants and antiseptics – Quantitative non-porous surface test without mechanical action for the evaluation of virucidal activity of chemical disinfectants used in the medical area – Test method and requirements (phase 2/step 2)
| 2024 Proficiency Testing Program | |
| Method | ID |
| EN 16777 | PT-16777-2.2-0924 |
Our products DILUTED SOFT SOAP

SHAPYPRO manufactures soft soap diluted used in the standards EN 1499:2013 Hygienic handwash “Test method and requirements (phase 2/stage 2)” and EN 1500:2013 Hygienic handrub “Test method and requirements (phase 2/stage 2)” for the preparation of the volunteer’s hands. As well as the one used for the procedure for Hygienic handwash reference (RP) for the EN 1499 standard.
COMPONENTS:
- Linseed oil: 50,0 parts by weight.
- Potassium hydroxide: 9,5 parts by weight.
- Ethanol (min. 95 %): 7,0 parts by weight.
- Hot distilled water: (75 ± 5) ºC: weight as needed.
Quality control of the soft soap.
We perform the quality control of the soft soap as indicated in EN 1499 Hygienic handwash “Test method and requirements (phase 2/stage 2)” and EN 1500 Hygienic handrub “Test method and requirements (phase 2/stage 2)” at Annex D in both.
| Quality control of the soft soap | |
|---|---|
| Determination | Result / Unit |
| Identity | Characteristic form according to Annex D |
| Purity | Clear liquid |
| Insoluble substances | < 5 mg |
| Free alkali, free acid | < 0,1 ml HCL o < 0,1 ml de NaOH |
| Loss on drying | < 45,0 % |
| Determination of content | 45,0 % – 50,0 % |
All products are supplied with safety data sheet, technical data sheet and certificate of analysis.
Please ask us for a non-binding quotation.
SHAPYPRO intercomparison programs

If you are accredited in the above-mentioned standards or are thinking of doing so and need to perform an intercomparison, Shapypro can help you. Do not hesitate to contact us.


