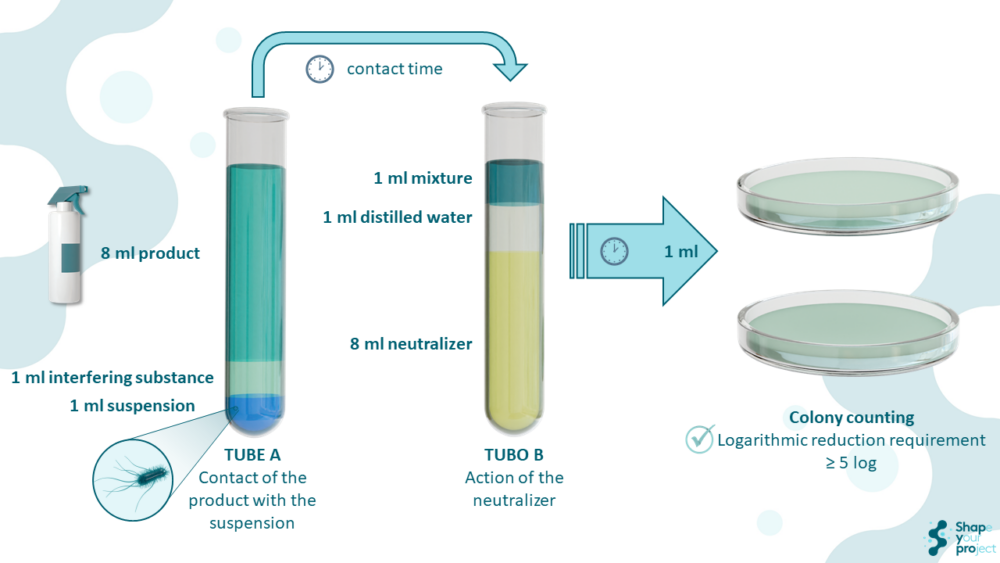
The quantitative suspension test is a methodology used to measure the ability of a disinfectant or antiseptic product to reduce the number of viable bacteria in a liquid suspension under specific conditions. This method is fundamental to ensuring that products meet the necessary efficacy requirements for safe and effective use.
This article will compare several key aspects of these standards, including their scope of application, test conditions, test organisms used, minimum logarithmic reduction requirements, and validation procedures. Through this comparison, the goal is to provide a clear and understandable view of the similarities and differences between these important standards.
Quantitative suspension test for disinfectant
The quantitative suspension test for disinfectant is a laboratory technique designed to evaluate the biocidal effectiveness of antiseptic and disinfectant products. It involves adding a specific amount of the product to a bacterial suspension and, after a defined contact time, neutralizing the disinfectant’s action and determining the number of surviving bacteria. This method is crucial because it simulates practical use conditions and provides quantitative data on the product’s ability to reduce bacterial load.
Test conditions include factors such as temperature, contact time, and the presence of interfering substances. These factors are selected to reflect the conditions under which the products will be used in practice. Interfering substances, such as bovine albumin and milk, are used to simulate the presence of organic matter that could affect the disinfectant’s effectiveness.
Scope of application
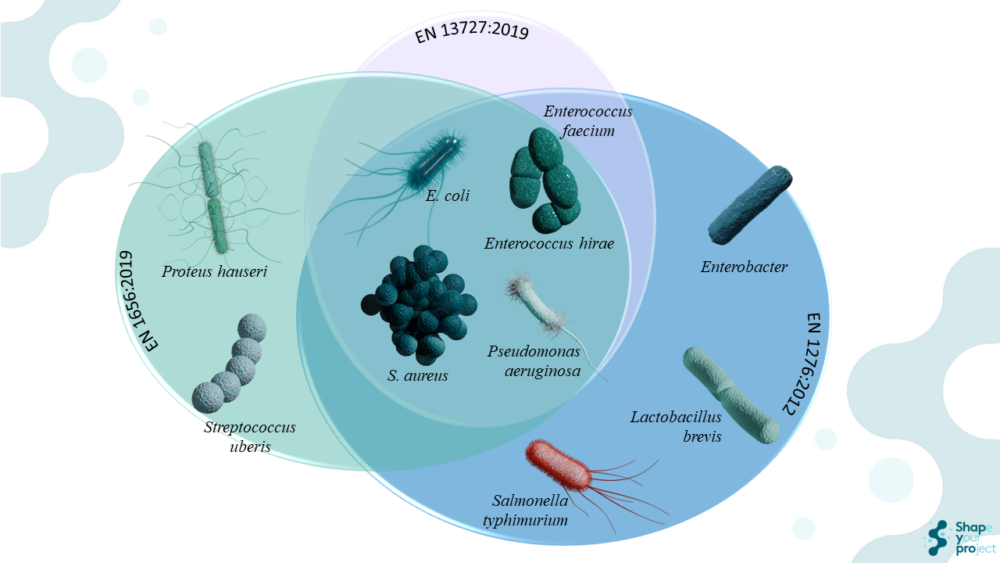
The standards EN 1656:2019, EN 1276:2019, and EN 13727:2012+A2:2015 apply to different contexts, although all share the objective of evaluating the bactericidal efficacy of chemical disinfectants and antiseptics. EN 1656:2019 focuses on evaluating the bactericidal activity of products used in the veterinary field. In contrast, EN 1276:2019 assesses bactericidal activity in food, industrial, domestic, and institutional areas, while EN 13727:2012+A2:2015 is specifically designed for the medical field, evaluating the bactericidal activity of products used in the disinfection of surfaces, hands, and medical equipment.
Conditions the suspension test for disinfectant
The conditions suspension test for disinfectant described in the standards are fundamental to ensuring that the results are comparable and reproducible. The standards specify clean and dirty conditions.
Clean conditions simulate situations where the surface to be disinfected is not significantly contaminated with organic matter. In EN 1276:2019, a 0.3 g/l bovine albumin solution is used, representing low levels of proteins that could interfere with the disinfectant’s action. On the other hand, dirty conditions simulate more challenging situations, where there is a higher amount of organic matter present. In these cases, a 3 g/l bovine albumin solution is used, representing a more contaminated environment that can hinder the disinfectant’s effectiveness. The standards consider the inclusion of additional relevant interfering substances depending on the specific application field, such as various proteins, lipids, and detergents.
In food contexts, the use of reconstituted milk at 1% (v/v) as an interfering substance is also specified in certain tests. Milk is relevant in the context of food processing, as it represents residues of dairy products that can be found on processing equipment and surfaces. Using milk in the tests ensures that disinfectants are effective under conditions that better mimic real situations in the food industry. To prepare this solution, guaranteed antibiotic-free and additive-free skimmed milk powder is reconstituted with 100 g of milk powder per liter of water, heated to high temperatures.
In EN 1656:2019, dirty conditions are described at two levels: low, using a 3 g/l bovine albumin solution, and high, combining a 10 g/l bovine albumin solution with 10 g/l yeast extract.
On the other hand, EN 13727:2012+A2:2015 details specific conditions for the medical field, including the use of bovine albumin and erythrocytes as interfering substances. To simulate dirty conditions in medical tests, a 3 g/l bovine albumin solution mixed with 3 ml/l of erythrocytes is used, reflecting the presence of blood and other proteins in the clinical environment. These conditions ensure that products are evaluated in a way that reflects their effectiveness in practical hospital use situations, where pathogen elimination is critical for preventing nosocomial infections.
Organisms on the suspension test for disinfectant
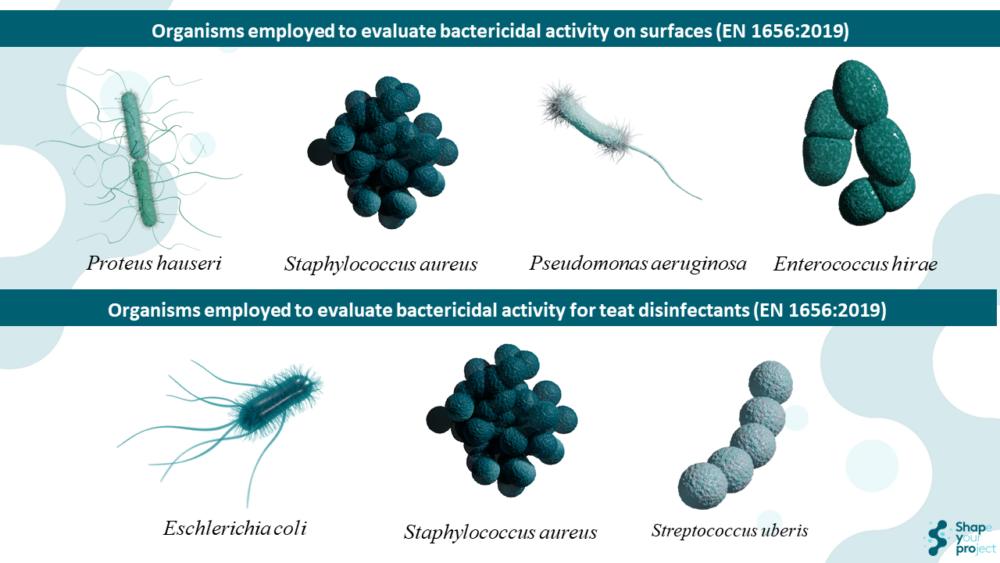
The test organisms used in these standards vary according to the field of application of the standard.
In EN 1656:2019, specific bacterial strains such as Enterococcus hirae ATCC 10541, Proteus hauseri ATCC 13315, Pseudomonas aeruginosa ATCC 15442, and Staphylococcus aureus ATCC 6538 are used for general disinfection products. For teat disinfectants, Escherichia coli ATCC 10536, Staphylococcus aureus ATCC 6538, and Streptococcus uberis ATCC 19436 are used. Enterococcus hirae is a Gram-positive bacterium used as a test organism due to its intrinsic resistance to many antimicrobial agents. Proteus hauseri, formerly known as Proteus vulgaris, is a Gram-negative bacterium relevant in the veterinary field. Pseudomonas aeruginosa is a Gram-negative bacterium known for causing infections in immunocompromised individuals and its resistance to disinfectants. Staphylococcus aureus is a Gram-positive bacterium that causes various infections, including skin infections and pneumonia. Escherichia coli is a Gram-negative bacterium found in the human and animal intestines. Streptococcus uberis is a Gram-positive bacterium relevant in mammary infections in the veterinary field.
EN 1276:2019, on the other hand, focuses on specific bacterial strains such as Pseudomonas aeruginosa ATCC 15442, Escherichia coli ATCC 10536, Staphylococcus aureus ATCC 6538, and Enterococcus hirae ATCC 10541. Additionally, Salmonella Typhimurium ATCC 13311, Lactobacillus brevis DSM 6235, and Enterobacter cloacae DSM 6234 can be used for specific applications. For temperatures above 40 °C, Enterococcus faecium ATCC 6057 should be used.
In the case of EN 13727:2012+A2:2015, relevant bacterial strains in the medical context are used, including Pseudomonas aeruginosa ATCC 15442, Staphylococcus aureus ATCC 6538, Escherichia coli K12 NCTC 10538, Enterococcus hirae ATCC 10541, and Enterococcus faecium ATCC 6057. The choice of these organisms is due to their clinical relevance and resistance to disinfectants, ensuring that products are effective against the pathogens that most commonly cause nosocomial infections.
If additional test organisms are used, they should be incubated under the optimal growth conditions specified in the test report, including temperature, time, atmosphere, and medium. These additional organisms must be verified to ensure their suitability and, if not classified in a reference center, must be adequately characterized. Furthermore, the testing laboratory or the national culture collection must maintain these organisms as a reference for a period of five years.
Minimum logarithmic reduction requirements and validation procedures
The standards establish minimum logarithmic reduction requirements to ensure that the products are effectively biocidal. Logarithmic reduction is a measure of the decrease in the number of viable microorganisms after treatment with a disinfectant.
In EN 1656:2019, products are required to achieve a reduction of at least 5 decimal logarithms under simulated low-level dirty conditions (3 g/l bovine albumin solution) or simulated high-level dirty conditions (10 g/l bovine albumin solution and 10 g/l yeast extract). For teat disinfectants, the minimum required reduction is also 5 decimal logarithms.
EN 1276:2019 and EN 13727:2012+A2:2015 require a reduction of at least 5 decimal logarithms for general applications, including surface disinfection and other non-hand specific uses. However, for products intended for handwashing, a reduction of 3 decimal logarithms is required.
Additionally, validation procedures are essential to ensure the reproducibility and reliability of the tests. All standards detail methods for the preparation of microorganism suspensions, incubation conditions, and calculations necessary to determine biocidal efficacy. The calculations include determining the number of colony-forming units (CFUs) before and after treatment with the disinfectant.
To do this, dilution-neutralization and membrane filtration methods are used to neutralize and eliminate biocidal activity before performing bacterial counts. These procedures ensure that test results adequately reflect the product’s effectiveness.
The dilution-neutralization method involves diluting the treated sample with a specific neutralizer that stops the disinfectant’s biocidal action, allowing only the surviving microorganisms to be counted. The membrane filtration method, on the other hand, involves filtering the treated sample through a membrane that captures the microorganisms, followed by washing with a rinse liquid that removes any disinfectant residue, allowing a precise count of the surviving microorganisms.
Comparative summary table for suspension test for disinfectant
| Aspect | EN 1656:2019 | EN 1276:2019 | EN13727:2012+A2:2015 |
| Objetive | Evaluation of bactericidal activity of disinfectant and antiseptic products | ||
| Test type | Suspension test | ||
| Scope of Application | Veterinary | Food, industrial, domestic, institutional | Medical |
| Test Conditions | Low-level dirty (3 g/l albumin) High-level dirty (10 g/l albumin + 10 g/l yeast extract) | Clean (0.3 g/l albumin) Dirty (3 g/l albumin) Milk (1% v/v) | Clean (0.3 g/l albumin) Dirty (3 g/l bovine albumin + 3 ml/l erythrocytes) |
| Test Organisms | Surface disinfectants
Pseudomonas aeruginosa, Staphylococcus aureus Enterococcus hirae, Proteus hauseri. Teat disinfectants Escherichia coli, Staphylococcus aureus, Streptococcus uberis |
Pseudomonas aeruginosa, Escherichia coli, Staphylococcus aureus, Enterococcus hirae, Enterococcus faecium | |
| Additional Test Organisms | Additional test organisms may be tested | Salmonella typhimurium, Lactobacillus brevis, Enterobacter cloacae | Additional test organisms may be tested |
| Logarithmic Reduction Requirements | General: ≥ 5 log
Teat disinfectants: ≥ 5 log |
General: ≥ 5 log
Handwashing: ≥ 3 log |
|
In summary, the standards EN 1656:2019, EN 1276:2019, and EN 13727:2012+A2:2015 provide a robust framework for evaluating the efficacy of antiseptics and disinfectants in various applications. Understanding the similarities and differences between these standards is essential for their correct application and to ensure the safety and effectiveness of products in different contexts.
The proper implementation of these standards not only guarantees the effectiveness of the products but also protects public health by ensuring that the disinfectants and antiseptics used in different sectors are suitable for their specific purposes.
Why Choose SHAPYPRO Proficiency Tests?
With over 10 years of experience in evaluating the efficacy of disinfectant products, we offer proficiency testing programs meeting ISO/IEC 17043 requirements in accordance with EN 14885:2022/AC:2023 standards.
Our extensive knowledge allows us to assist clients in developing new methods and R&D projects. We have developed proficiency testing programs under EN 1656:2019, EN 1276:2019, and EN 13727:2012+A2:2015 standards previously.
We are manufacturers of the necessary diluted soft soap for both developments. We offer custom proficiency testing.
The tests are conducted following validated international standards and guidelines.
Would you like to get our 5-year agenda? Write to us!


