What is the purpose of EN 14885?
This European Standard to group the European Standards to which products have to conform in order to support the claims for microbicidal activity which are referred to in this European Standard.
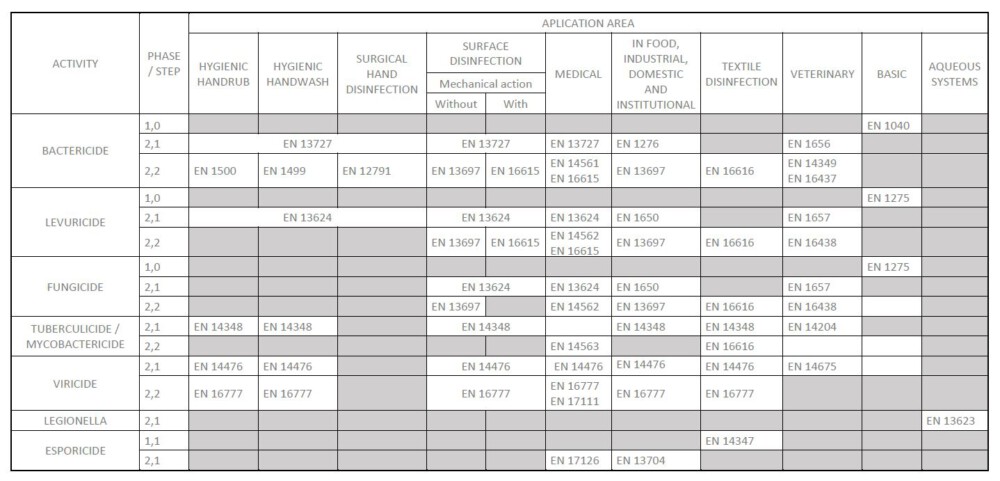
What kind of standards are covered by this regulation and for whom is it intended?
It is applicable to products for which activity is claimed against the following microorganisms: vegetative bacteria (including mycobacteria and Legionella), bacterial spores, yeasts, fungal spores and viruses (including bacteriophages). It is intended to:
- enable manufacturers of products to select the appropriate standards to be used in order to provide data which support their claims for a specific product;
- enable testing biocidal efficacy labs of the product to group the standards families in their accreditation scope ISO/IEC 17025 in order to preparer the proficiency testing program they will attended in relation to the use for which they intend to use the product;
- assist regulatory authorities in assessing claims made by the manufacturer or by the person responsible for placing the product on the market. As also, assist accreditation bodies to check the grouping by families in the scope of accreditation of analytical laboratories.
What areas of application are covered by the standard?
It is applicable to products to be used in the area of human medicine, the veterinary area and in food, industrial, domestic and institutional areas.
Human Medicine Area
In the area of human medicine, it is applicable to chemical disinfectants and antiseptics to be used in areas and situations where disinfection or antisepsis is medically indicated. Such indications occur in patient care, It is intended to:
- in hospitals, in community medical facilities and dental institutions;
- in clinics of schools, of kindergartens and of nursing homes;
- and may also occur in the workplace and in the home. It may also include services such as in laundries and kitchens supplying products directly for the patient.
Veterinary Area
In the veterinary area it is applicable to chemical disinfectants and antiseptics to be used in the areas of:
- breeding;
- husbandry;
- veterinary care facilities;
- production, transport and disposal of animals.
It is not applicable to chemical disinfectants used in the food chain following death and entry to the processing industry.
Food, Industrial, Domestic and Institutional Areas.
In food, industrial, domestic and institutional areas it is applicable to chemical disinfectants and antiseptics to be used in:
- processing;
- distribution and retailing of food of animal or vegetable origin;
- It is also applicable to products for all public areas.
It is not applicable to public areas where disinfection is medically indicated (homes, catering, schools, nurseries, transports, hotels, offices etc.) and products used in packaging, biotechnology, pharmaceutical, cosmetic etc. industries.
does SHAPYPRO currently have an proficiency testing program open on any of these standards?
Yes, we are open registration time in PT program according to EN 14476 Chemical disinfectants and antiseptics – Quantitative suspension test for the evaluation of virucidal activity in the medical area – Test method and requirements (Phase 2/Step 1)

