Quality is paramount in our Proficiency Testing Program. We strive to uphold the highest standards in every aspect of Intercomparison quality. Get ready to showcase your excellence and contribute to the quality that defines us!
Ensuring quality in intercomparisons is crucial for obtaining accurate and reliable results. Here are some measures that can be taken to ensure quality:
- Establish clear participation criteria: Clearly define who can participate in ring trials programs and what requirements they must meet to ensure data quality.
- Select representative participants: Ensure the necessary number of participants.
- Reference standards and quality control materials: Use items that guarantee homogeneity and stability.
- Planning and organization: Establish a detailed plan for the execution of the intercomparisons, including sample distribution, analysis methods, deadlines, and communication among participants.
- Statistical analysis: Use appropriate statistical techniques to analyse the data collected during the ring trials and evaluate the accuracy and precision of the results.
- Uncertainty evaluation: Establish validated methods for uncertainty evaluation in the ring trials programs.
- Feedback and continuous improvement: Provide feedback to participants on their results and seek opportunities to improve processes and procedures in future intercomparisons.
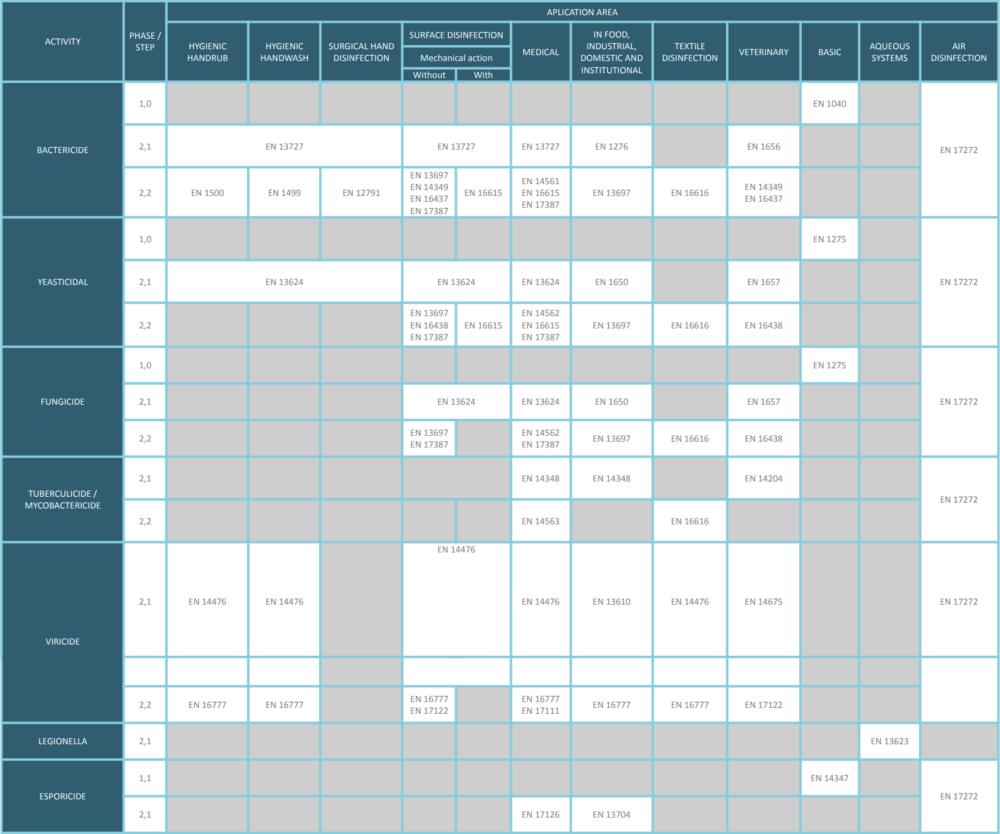
By following these measures and maintaining a rigorous approach in the planning, execution, and analysis of intercomparisons, the quality of the results obtained can be ensured. In our 2024 agenda we have published a summary table from EN 14885.
What do we do with invalid data from our participants?
- Internal investigation: Thoroughly analyses the invalid results and determine whether they are due to human errors, technical issues, or any other reason related to the proficiency testing process. It is important to identify the underlying cause of the problem.
- Communication with the participant: Contact the participant who provided the invalid results to discuss the situation. Provide constructive feedback on the results and request a detailed explanation of the procedures used and any issues faced during the proficiency testing process.
- Data revaluation: Evaluate whether it is possible to correct the invalid results or if they need to be completely discarded from the proficiency testing process. In some cases, it may be possible to work with the participant to correct the errors and obtain valid results that meet the standards of the proficiency testing process.
- Review of procedures and policies: Evaluate whether the current procedures and policies of the proficiency testing process are adequate to prevent the occurrence of invalid results in the future. Additional quality control measures or training may be implemented to avoid similar issues in future proficiency testing processes.
- Corrective actions: In severe cases of invalid results due to negligence or misconduct during the proficiency testing process, corrective actions may be applied according to the organization’s policies or the proficiency testing process.
- Transparency and communication: Keep all participants and stakeholders informed about the situation transparently and honestly during the proficiency testing process. This can help preserve trust in the process and demonstrate commitment to the quality and integrity of the process.
- Monitoring and continuous review: Regularly monitor the performance of participants and continuously review the procedures of the proficiency testing process to ensure that quality and reliability are maintained over time in the process.
Ultimately, it is essential to address invalid results diligently and take necessary measures to maintain the quality and credibility of the proficiency testing process.
What do we do with invalid data from our participants?
The quality of test items is essential to ensure the validity, reliability, and relevance of any assessment. Here are some important aspects to consider ensuring the quality of test items:
- Validity: Test items should accurately measure the knowledge or skills intended to be assessed. It’s important for items to align with program objectives and reflect appropriate content and difficulty levels.
- Reliability: Test items should be consistent and reliable in their ability to measure the same construct each time they are used.
- Clarity and coherence: Test items should be clear and precise to avoid confusion or misinterpretation by participants. Additionally, items should be consistent in their structure and format to facilitate understanding and response by participants.
When it comes to intercomparisons, the homogeneity and stability of test items are crucial to ensure consistency and reliability of the obtained results.
Homogeneity: Homogeneity refers to the extent to which test items evaluate the same skill or concept consistently. In the context of intercomparisons, test items must be homogeneous so that all participants are being evaluated on the same skills or competencies. This means that items should be clear, relevant, and aligned with the objectives of the intercomparison. Furthermore, the difficulty and discrimination of items should be similar for all participants, ensuring that the assessment is fair and equitable.
Stability: Stability refers to the consistency of results obtained over time or under different conditions. In the context of intercomparisons, test items must be stable so that results are reliable and consistent over time and across different groups of participants. This implies that items should be robust enough to not be affected by external factors or variations in administration conditions. Additionally, items should be able to measure the same skill or concept consistently at different times or contexts.
To ensure the homogeneity and stability of test items in intercomparisons, it’s important to conduct a rigorous process of item design and review. This may include the involvement of subject matter experts, conducting pilot tests to assess item quality, and regular review of items to ensure their relevance and accuracy. Additionally, it’s important to conduct statistical analyses to evaluate the homogeneity and stability of items and make adjustments as necessary. Ultimately, maintaining the homogeneity and stability of test items is essential to ensure the quality and reliability of intercomparisons and the obtained results.
Our ongoing Intercomparison quality program
EN14675 Quantitative suspension test for the evaluation of the virucidal activity of chemical antiseptics and disinfectants used in the veterinary area (phase 2 / stage 1).
EN16777 quantitative non-porous surface test without mechanical action for the evaluation of the virucidal activity of chemical disinfectants used in medicine (phase 2 / step 2).
Ask us a question by following this link:
If you are accredited in the above-mentioned standards or are thinking of doing so and need to perform an intercomparison, Shapypro can help you. Do not hesitate to contact us.


